Our Technology
NAAmatrix™ a bioresorbable natural-amino-acid matrix engineered for safety, scalability, and long-acting ocular drug delivery
Safe by design. Durable by nature
A new class of drug‑delivery that turns existing and pipeline retinal drugs into long‑acting therapies
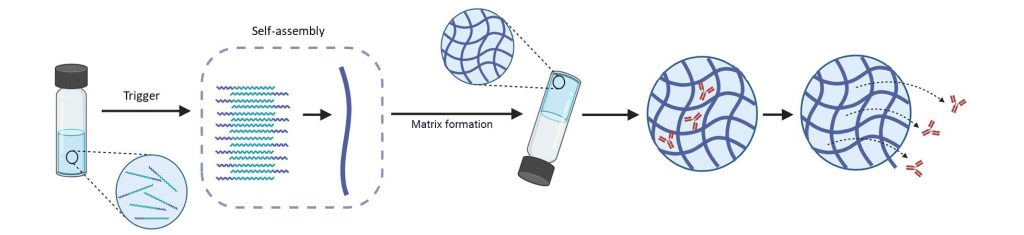
Key Features Of The NanOptima’s NAAmatrix™ Drug Delivery Platform
Turns existing retinal drugs into long-acting therapies
- Long-acting delivery & dosing beyond six months from a single intravitreal injection
- Tunable release kinetics – customizable for duration, release rate, and dose
- Safety by design – demonstrated in vivo tolerability with clean histopathology
- Biocompatible and fully bioresorbable – high water content (~96%), degrades into natural amino acids
- Broad drug compatibility across leading anti-VEGF biologics (aflibercept/Eylea® // bevacizumab/Avastin®), novel peptides, and small molecules, without drug modification, unlocking value through long-acting pipeline formulations across current and future portfolios and retinal indications
- Optically transparent, pH-neutral and non-inflammatory by design to be well tolerated
- Scalable manufacturing - stable at 4°C and suitable for scalable sterile fill–finish manufacturing
- Attractive partnering profile – offers lifecycle extension and differentiation for incumbent retinal brands while maintaining familiar modes of administration and manufacturing routes
No surgery, devices, implants, synthetic hydrogels/PLGA, curing, new equipment, or retraining required.
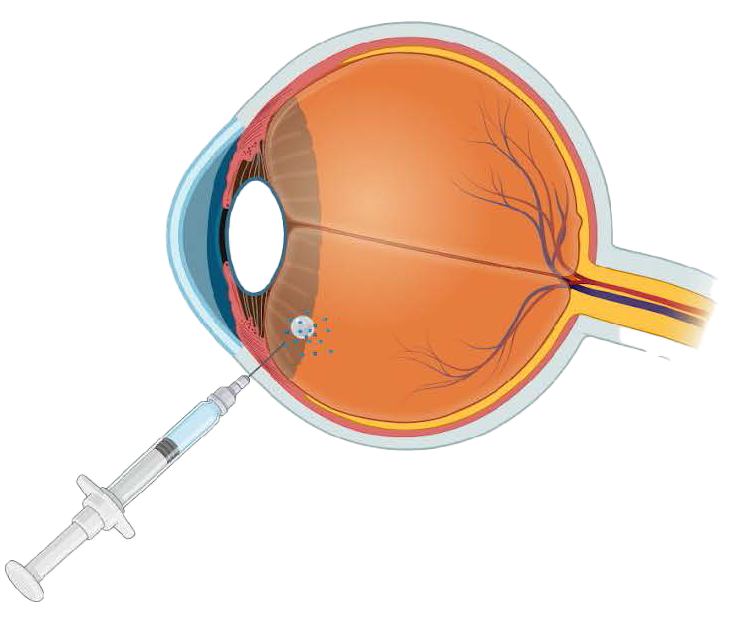
Clinical And Commercial Advantages
Designed to fit seamlessly into existing retinal care and commercial infrastructure.
- Lifecycle extension for leading assets - enables long-acting line extensions of established retinal biologics without changing the active molecule
- Differentiation vs. competitors and biosimilars - clear value proposition around fewer injections and sustained control, while leveraging familiar brands.ents significantly reduce the number of annual injections required by patients whilst also significantly reducing the number of appointments clinicians are required to schedule.
- Minimal CMC and device complexity – no implants, devices, or drug conjugation; designed to fit within standard biologics manufacturing and fill–finish workflows
- Frictionless for rapid clinical adoption – same procedure, administered via a standard 30G intravitreal injection, with no change in procedure or clinic workflow
- Reduces clinic burden – by extending treatment beyond six months, has the potential to relieve injection bottlenecks and free up capacity in busy retina clinics
Patient advantages
Designed around what matters most to patients living with chronic retinal disease.
- Fewer injections, fewer visits - support better quality of life – extended release beyond six months is designed to reduce treatment burden (travel, time off work, and caregiver reliance)
- Improved real-world adherence – extended dosing intervals are designed to reduce missed appointments, under-treatment, and associated vision loss
- Reduce under-treatment – sustained drug exposure is intended to reduce peaks and troughs of current therapies, and loss of disease control driven by missed or delayed appointments
- No risks associated with permanent implants, surgery, synthetic materials – fully bioresorbable NAAmatrix™ that degrades into natural amino acids
Focus on improved long-term vision – by lowering treatment burden and improving adherence, the platform is designed to help patientsstay on therapy and preserve long-term visual outcomes.
Together, we can drive transformative progress in ophthalmic care.
If you are interested in exploring partnership opportunities or learning more about our technology, we invite you to connect with us via the contact form below.
